Switching off a memory
The decoder said “Room A.” The HSE detector cleared it. The laser fired. What actually happened inside the hippocampus in the next few hundred milliseconds? This chapter is about the biology on the other side of that TTL pulse — how light silences a neuron, why silencing some neurons makes other neurons fire harder, and why the net effect on memory is disruption rather than random noise.
The tool is archaerhodopsin-T, or ArchT. It is a light-sensitive proton pump found originally in soil archaea, re-engineered for neuroscience use. When a neuron expressing ArchT is illuminated with yellow-green light at roughly 532 nanometres, the pump activates and pushes positive charge out of the cell. The membrane potential drops. Spiking stops. The effect begins within a millisecond of illumination and reverses within a few milliseconds of the light switching off. For a closed-loop system operating at the timescale of a single sharp-wave ripple, that response speed is exactly what the experiment needs.
Delivering ArchT to the target cells requires a viral vector. An adeno-associated virus carrying the ArchT gene under the CaMKIIα promoter is injected into CA1 of both hippocampi; the promoter is active only in excitatory cells, so expression is pyramidal-cell specific. A green-fluorescent protein tag (the “T” in ArchT-T) co-localises with the opsin, providing a fluorescent readout of which cells expressed the virus — the histology check that every such experiment requires before any data can be interpreted. Four to six weeks after injection, expression is stable and the animal can be implanted.
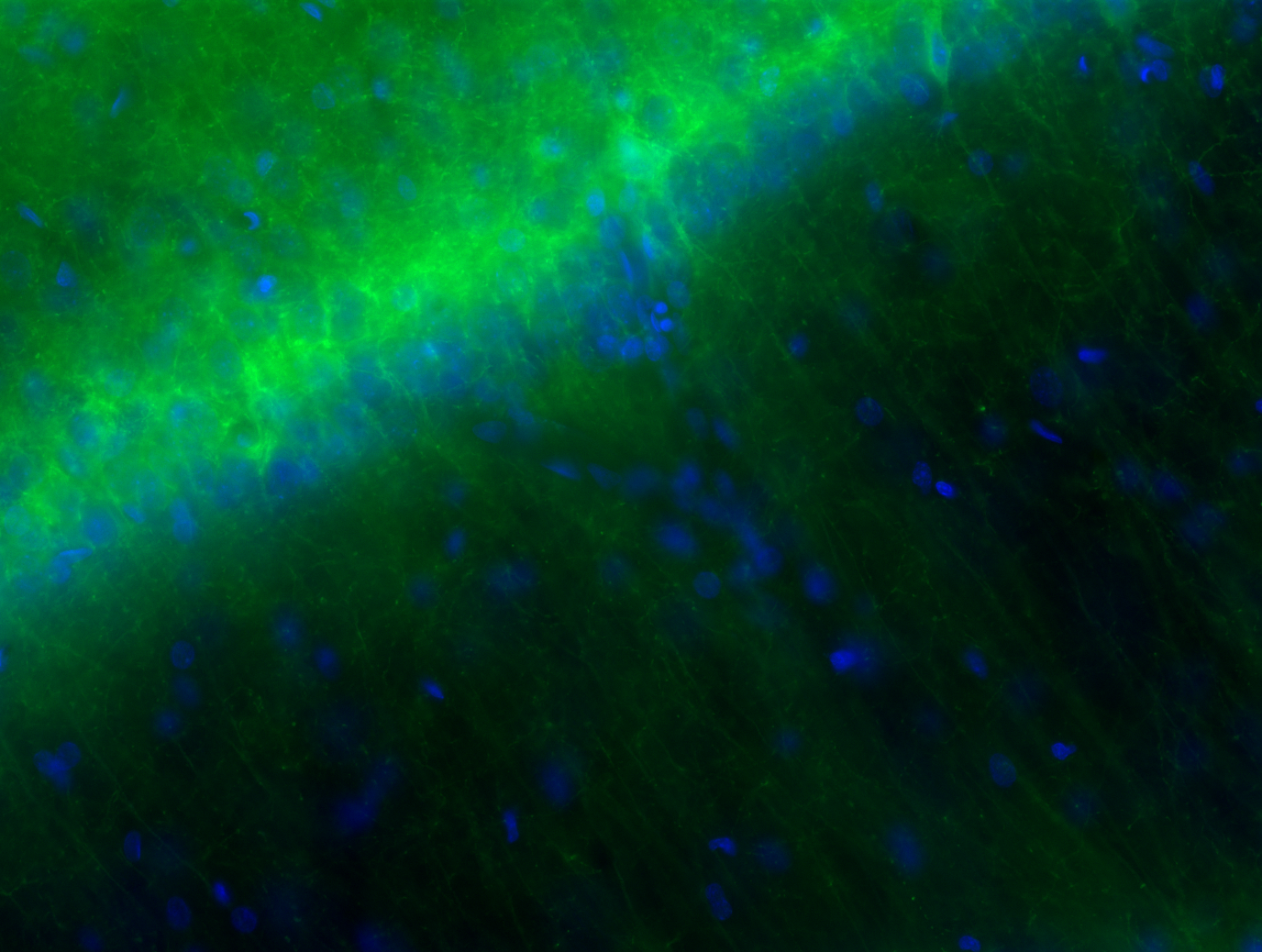
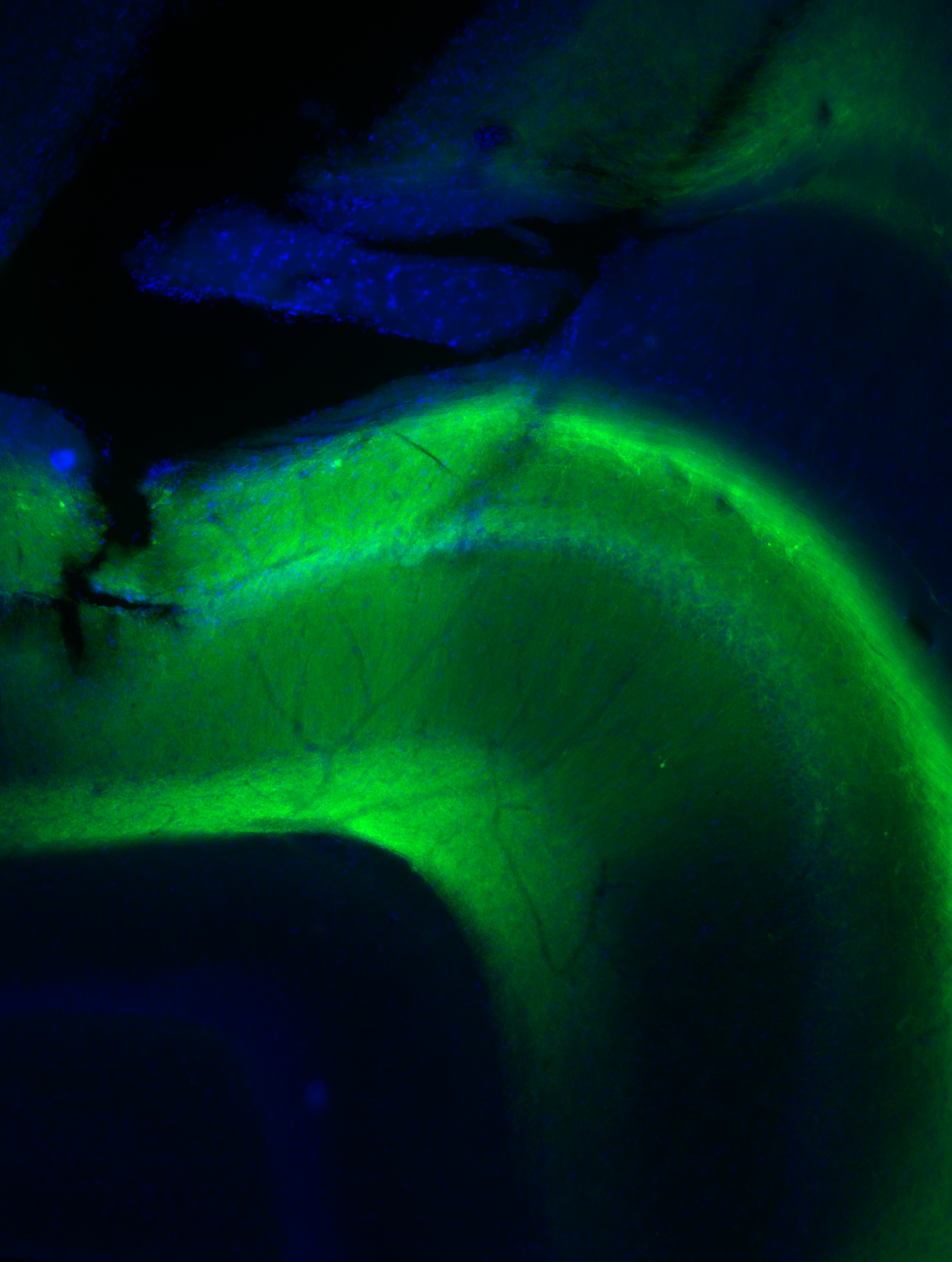
The circuit: why silencing produces excitation
Here is the part that trips people up. Silencing a population of pyramidal cells does not simply remove activity from the hippocampus. It also, transiently, increases activity in other pyramidal cells.
The reason is the local interneuron circuit. CA1 pyramidals drive recurrent inhibition through a set of fast-spiking interneurons — principally parvalbumin-positive basket cells — that in turn suppress the broader pyramidal population. This feedback keeps the network from cascading into synchronous bursts at every moment. When a subpopulation of pyramidals (the ArchT-expressing ones) is silenced, their excitatory drive to those interneurons drops. The interneurons receive less input, fire less, and therefore provide less inhibition to the remaining pyramidals. Those remaining cells — the ones that do not express ArchT — briefly lose their tonic suppression and fire harder.
This is disinhibition, and it is not a bug unique to this experiment; it is a generic feature of any circuit in which excitatory cells drive their own inhibitors. The animation below shows the sequence. Watch the order: ArchT cells dim first, then interneurons quiet, then the non-ArchT pyramidals brighten. The whole cascade unfolds in the same hundred-millisecond window as the disrupted SWR.
The disinhibition raises a legitimate concern: if non-ArchT pyramidals fire harder during the laser pulse, does the intervention strengthen parts of the replay pattern rather than simply disrupting it? The paper addressed this directly. The place-cell identity of the disrupted and non-disrupted populations was tracked across sessions, and the memory effect — the reduction in probe performance specifically for the target room — held across animals regardless of how much of the population happened to express ArchT. The dominant effect is ensemble-level disruption, not selective reinforcement of the non-expressing fraction.
Why content-specific disruption is different
Earlier closed-loop sleep-disruption experiments (Girardeau et al. 2009, Ego-Stengel and Wilson 2010) established that shutting off SWRs during post-task sleep impairs spatial memory. Those experiments used electrical stimulation that interrupted every ripple, regardless of content. The design is powerful but coarse: you cannot conclude from a memory deficit whether replay was driving consolidation or merely correlating with it, because you have disrupted all replay indiscriminately. Any control memory the animal might have is equally disrupted.
Content-specific disruption changes the logical structure of the experiment. If a rat trained on two arenas shows a deficit in Room A memory but intact Room B memory after selective Room A replay disruption — with both measured in the same animal, from the same neurons, across the same sleep session — then the result is internally controlled in a way that blanket disruption cannot achieve. Room B memory is the counterfactual: what the animal’s brain would have done to Room A if the laser had stayed off.
The disinhibition adds texture to that claim. The non-ArchT pyramidals experience a brief excitation during each targeted SWR. If anything, this creates a mild bias toward strengthening Room B cells (if they happen to share firing with the non-ArchT fraction in Room A), which would work against finding a deficit. The fact that Room A probe performance drops despite this counter-pressure makes the result, if anything, a conservative estimate of the disruption’s actual magnitude.
The biology and the engineering are now fully assembled. Chapter 8 is the payoff: did the rat forget?